
Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation | Science

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Thiol–ene reactions leading to fatty acid based monomers for polyester... | Download Scientific Diagram
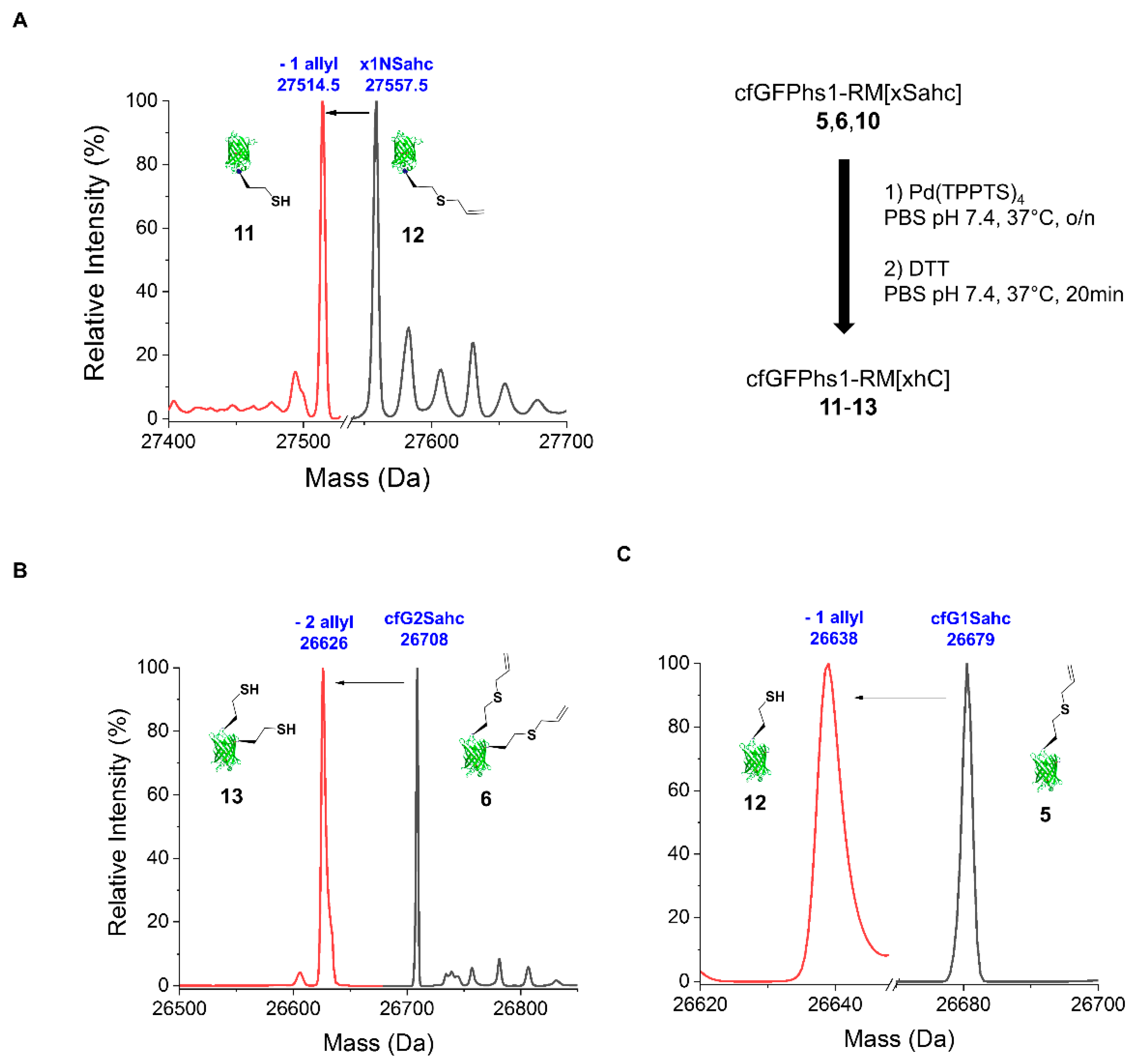
IJMS | Free Full-Text | In-Cell Synthesis of Bioorthogonal Alkene Tag S-Allyl-Homocysteine and Its Coupling with Reprogrammed Translation | HTML
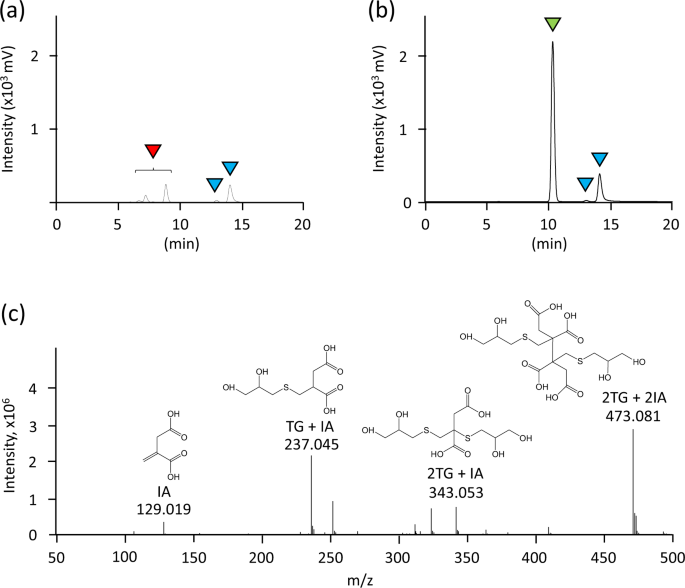
DISCOVER: A facile structure-based screening method for vinyl compound producing microbes | Scientific Reports
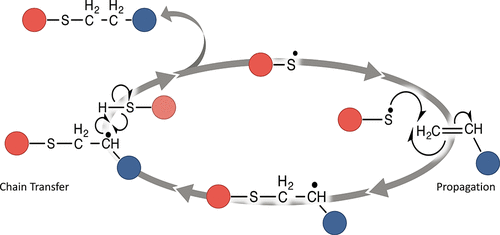
Effects of 1°, 2°, and 3° Thiols on Thiol–Ene Reactions: Polymerization Kinetics and Mechanical Behavior - Macromolecules - X-MOL
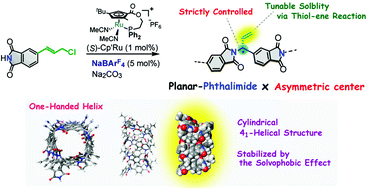
Synthesis of an optically active polymer containing a planar phthalimide backbone by asymmetric polymerization - Polymer Chemistry (RSC Publishing)
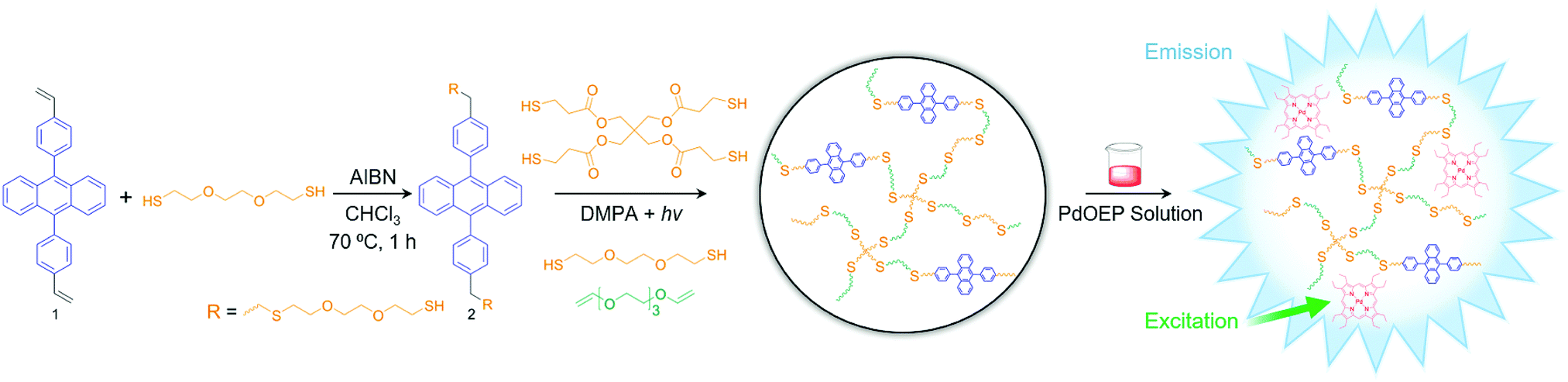
Thiol–ene click chemistry: a modular approach to solid-state triplet–triplet annihilation upconversion - Journal of Materials Chemistry C (RSC Publishing)

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

PDF) Versatile cross-linked fatty acid-based polycarbonate networks obtained by thiol–ene coupling reaction

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
![Organometallic Chemistry: Thiol–Ene “Click” Reaction Triggered by Neutral Ionic Liquid: The “Ambiphilic” Character of [hmim]Br in the Regioselective Nucleophilic Hydrothiolation Organometallic Chemistry: Thiol–Ene “Click” Reaction Triggered by Neutral Ionic Liquid: The “Ambiphilic” Character of [hmim]Br in the Regioselective Nucleophilic Hydrothiolation](http://3.bp.blogspot.com/-_0A3RB21QrE/VIABeiUhmeI/AAAAAAAALVA/CDamsXdd3zI/s1600/1.gif)
Organometallic Chemistry: Thiol–Ene “Click” Reaction Triggered by Neutral Ionic Liquid: The “Ambiphilic” Character of [hmim]Br in the Regioselective Nucleophilic Hydrothiolation

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
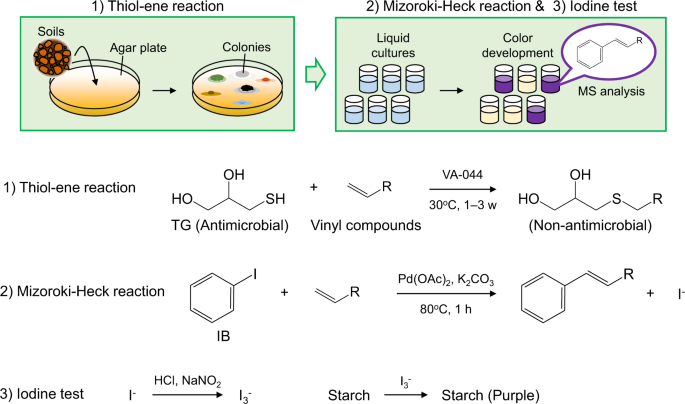
DISCOVER: A facile structure-based screening method for vinyl compound producing microbes | Scientific Reports
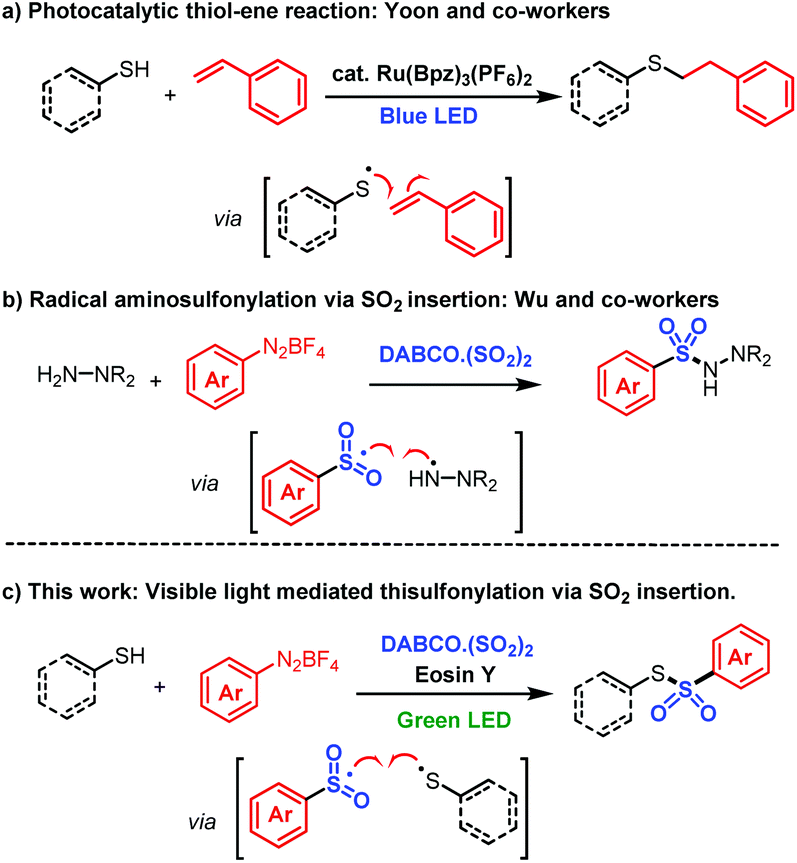
Visible-light mediated sulfonylation of thiols via insertion of sulfur dioxide - Organic & Biomolecular Chemistry (RSC Publishing)

Castor oil based UV-cured coatings using thiol-ene click reaction for thermal degradation with flame retardance - ScienceDirect







