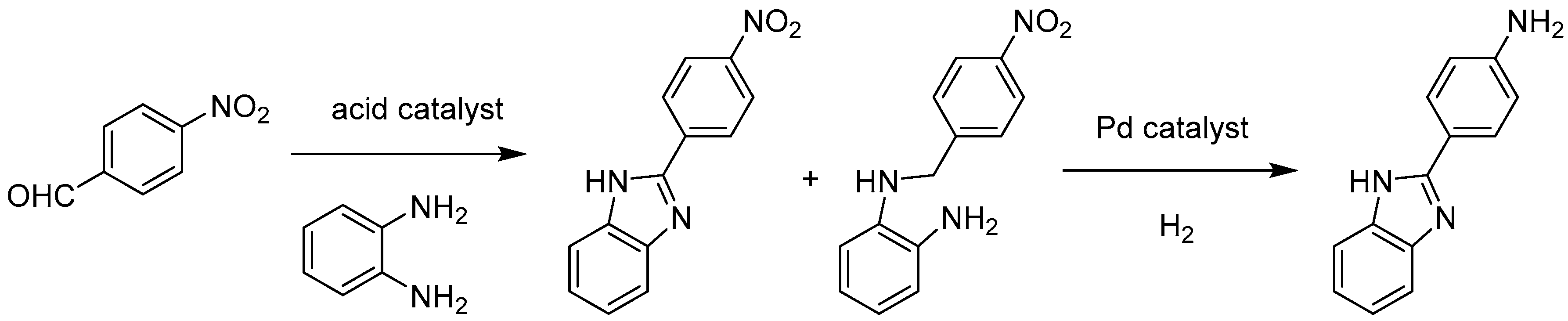
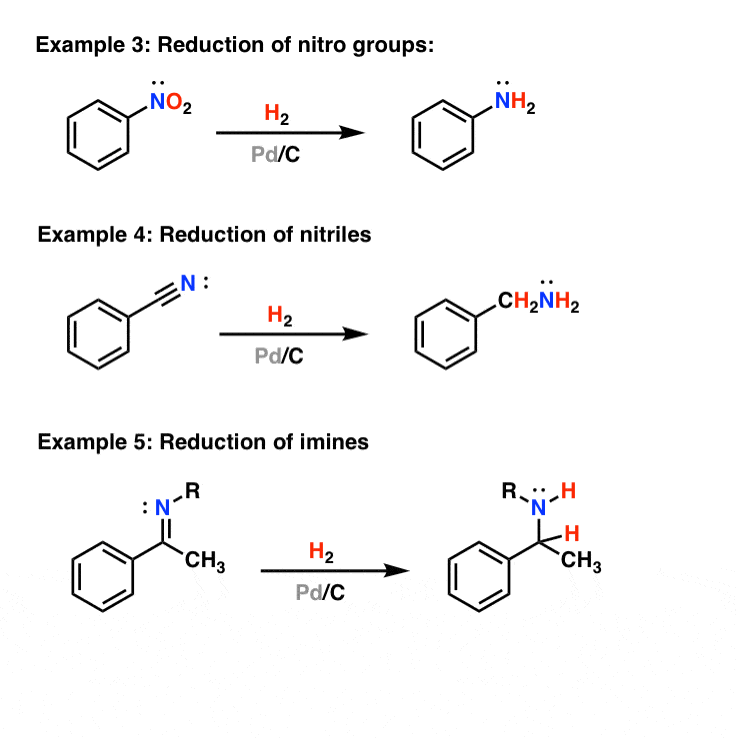
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
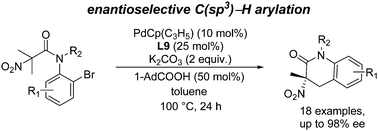
Asymmetric construction of quaternary α-nitro amides by palladium-catalyzed C(sp3)–H arylation - Chemical Communications (RSC Publishing)

Palladium(II) Acetate Catalyzed Reductive Heck Reaction of Enones; A Practical Approach - Mannathan - 2015 - ChemCatChem - Wiley Online Library

Proposed mechanism for transfer hydrogenation of nitroarenes over Pd/ZrP. | Download Scientific Diagram
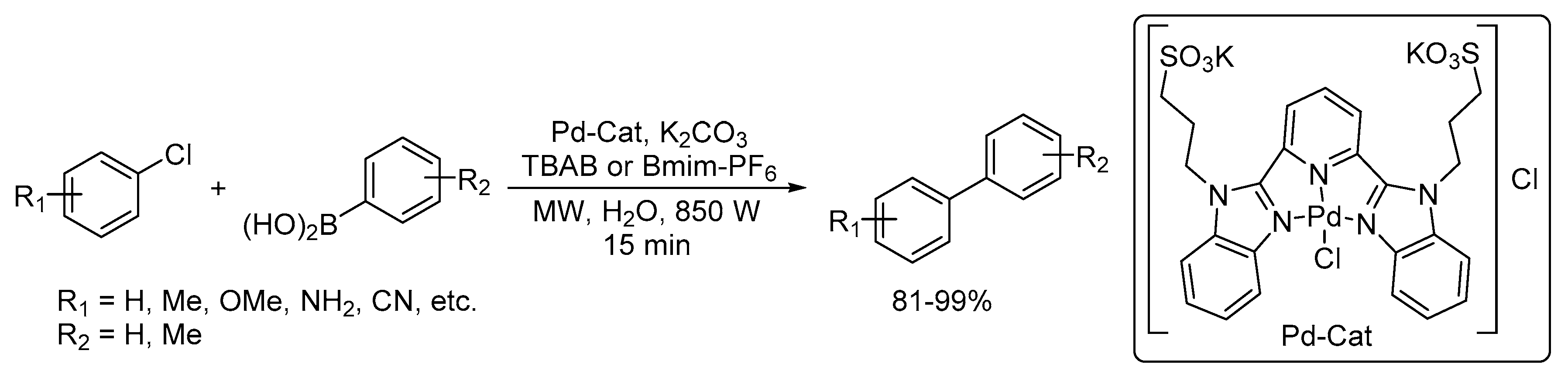
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Green synthesis & catalytic study of sucrose stabilized Pd nanoparticles in reduction of nitro compounds to useful amines - ScienceDirect
![PDF] Palladium-Catalyzed Silane/Siloxane Reductions in the One-Pot Conversion of Nitro Compounds into Their Amines, Hydroxylamines, Amides, Sulfonamides, and Carbamates | Semantic Scholar PDF] Palladium-Catalyzed Silane/Siloxane Reductions in the One-Pot Conversion of Nitro Compounds into Their Amines, Hydroxylamines, Amides, Sulfonamides, and Carbamates | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f73284494b8bfbabc67153e15e34aac110787a87/2-Table1-1.png)
PDF] Palladium-Catalyzed Silane/Siloxane Reductions in the One-Pot Conversion of Nitro Compounds into Their Amines, Hydroxylamines, Amides, Sulfonamides, and Carbamates | Semantic Scholar
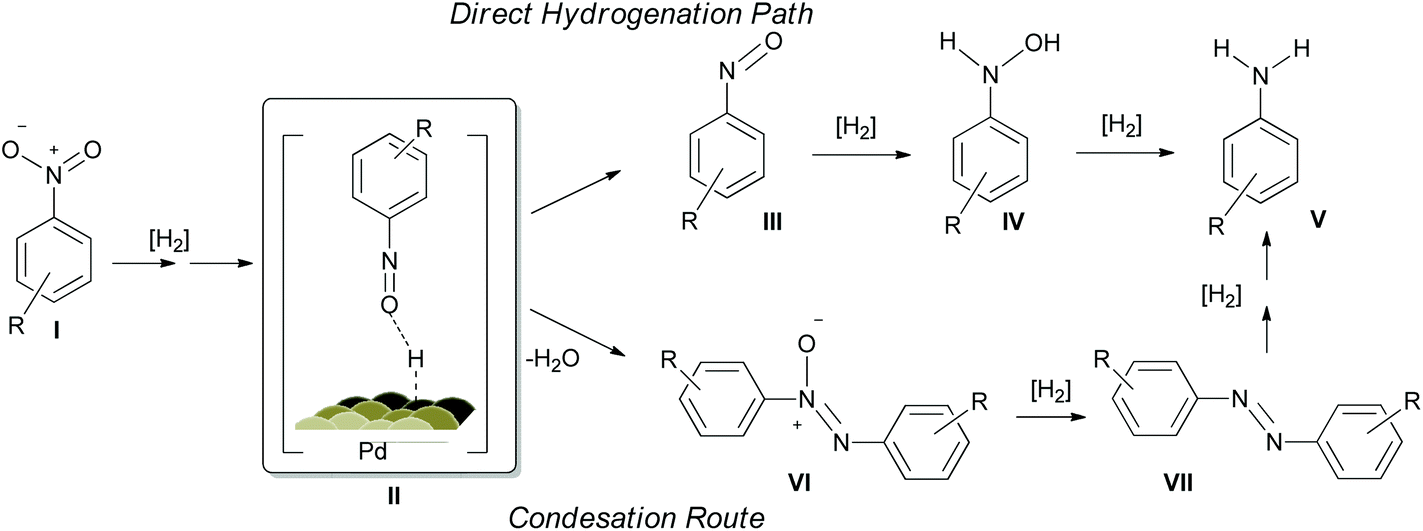
PVP-Pd nanoparticles as efficient catalyst for nitroarene reduction under mild conditions in aqueous media - Green Chemistry (RSC Publishing)
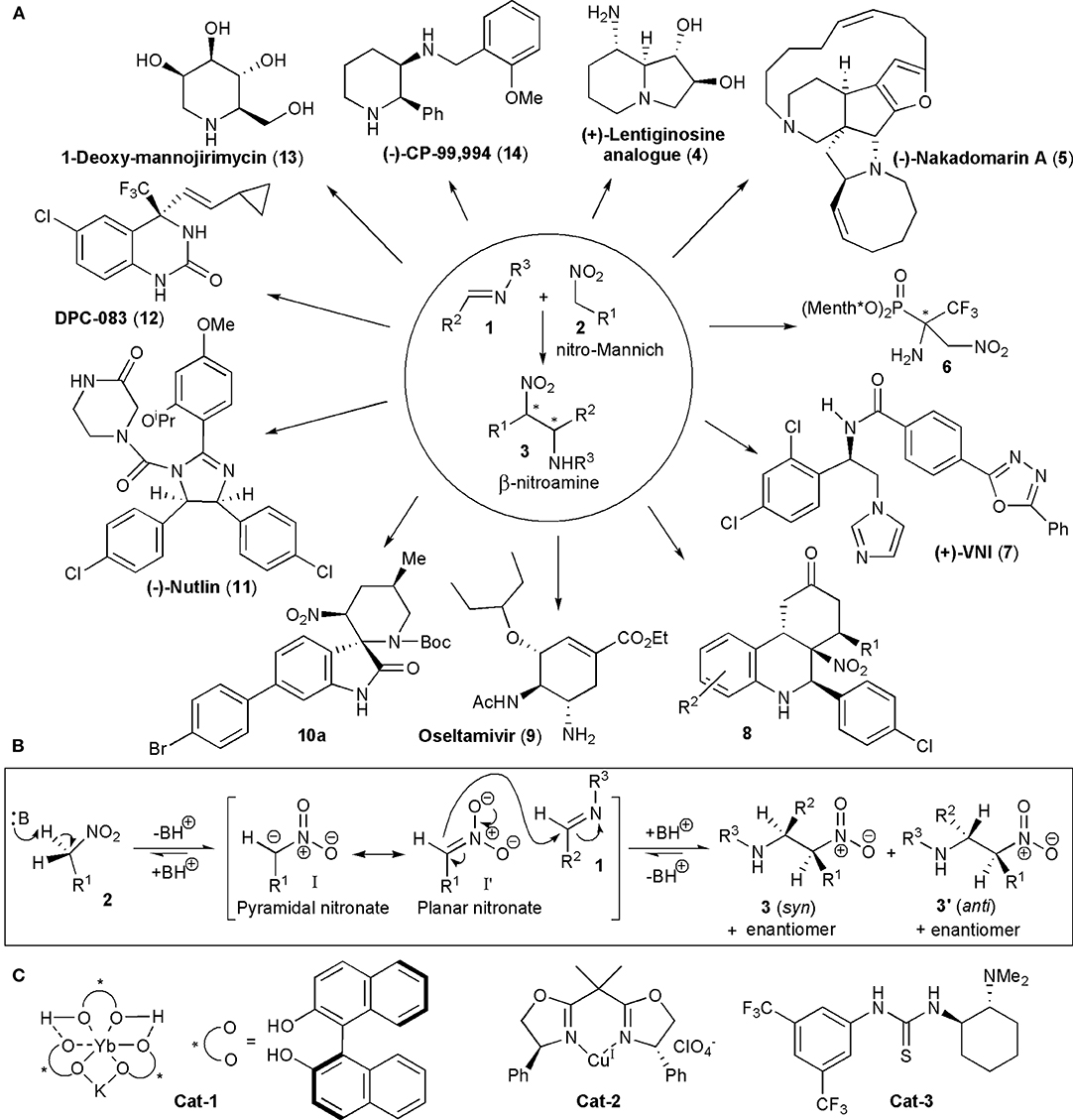
Frontiers | The Stereoselective Nitro-Mannich Reaction in the Synthesis of Active Pharmaceutical Ingredients and Other Biologically Active Compounds | Chemistry
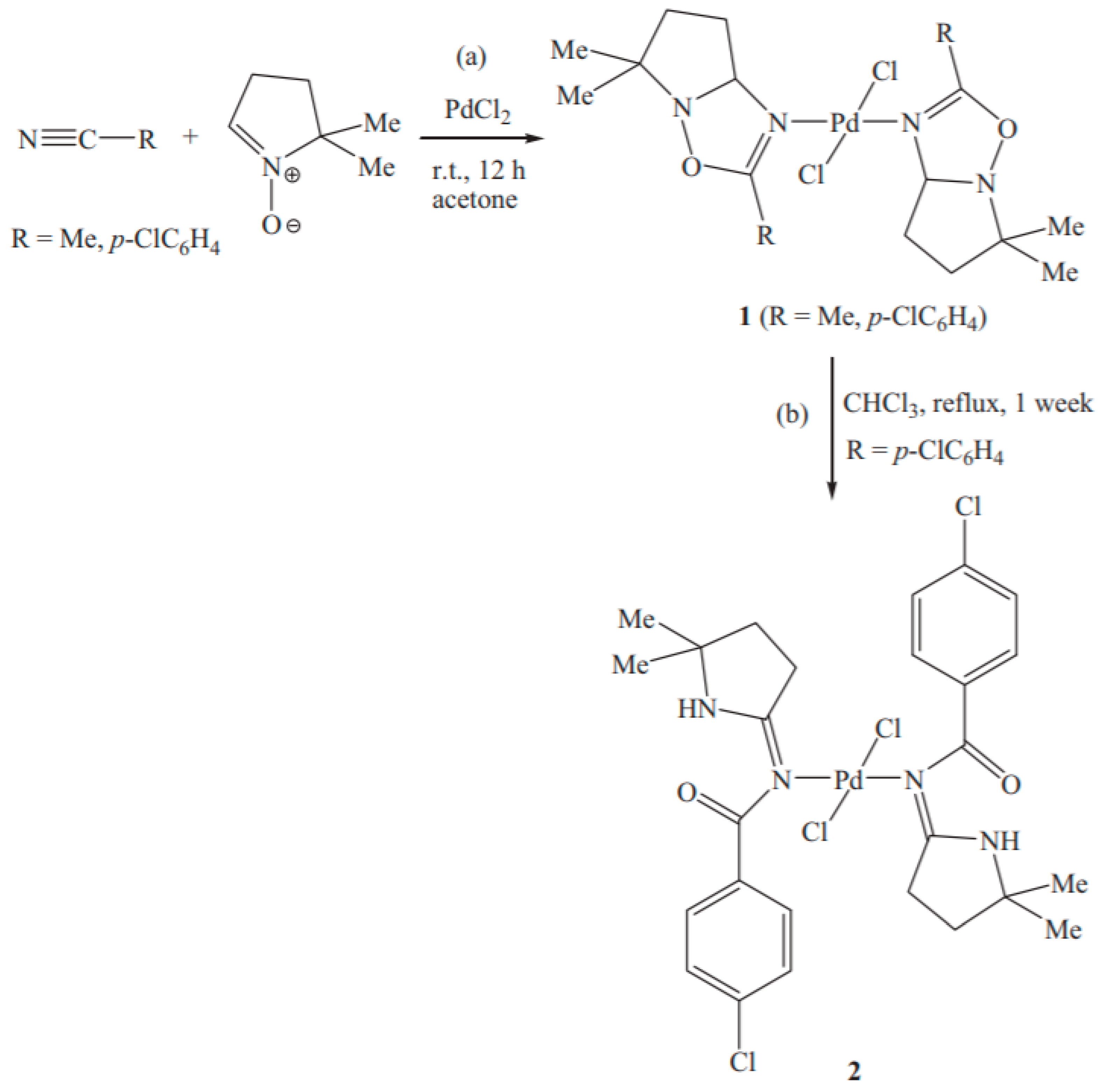
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML
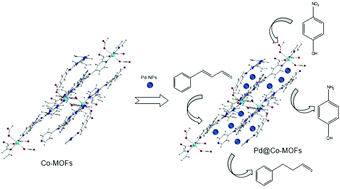
A new cobalt metal–organic framework as a substrate for Pd nanoparticles applied in high-efficiency nitro phenol degradation and cinnamaldehyde hydrogenation - Dalton Transactions (RSC Publishing)

Ligand characteristics and in situ generation of Pd active species towards CC coupling using series of 2-(1H-imidazol-2-yl)phenols - ScienceDirect

Table 6 from Palladium-Catalyzed Silane/Siloxane Reductions in the One-Pot Conversion of Nitro Compounds into Their Amines, Hydroxylamines, Amides, Sulfonamides, and Carbamates | Semantic Scholar
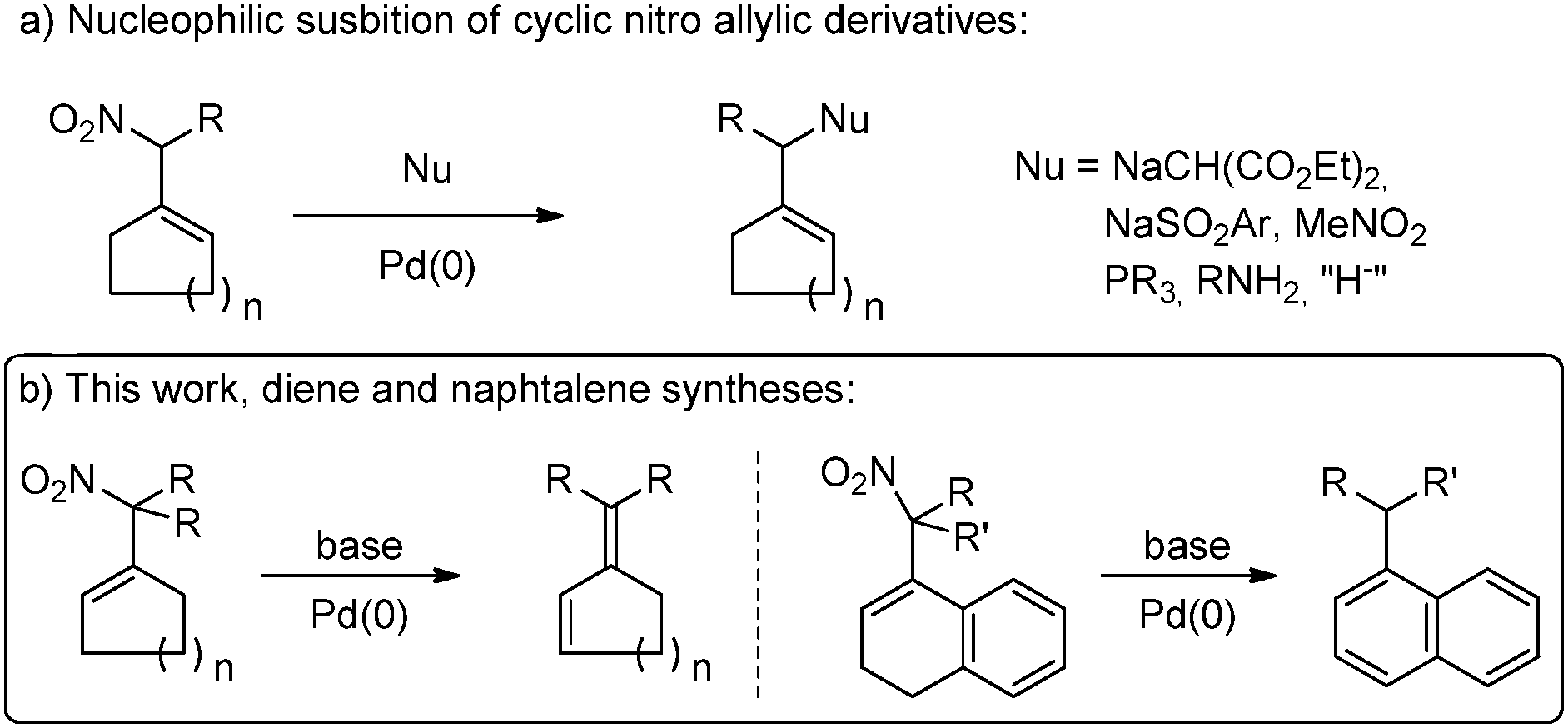
Palladium triggered diene formation from nitro allylic compounds: a versatile entry into naphthalene derivatives - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC06536E