N-Arylation of Amino Acid Esters to Expand Side Chain Diversity in Ketoxime Peptide Ligations. - J. Org. Chem. - X-MOL

PDF) Palladium-Catalyzed α-Arylation of Dimethyl Malonate and Ethyl Cyanoacetate with o-Alkoxybromobenzenes for the Synthesis of Phenylacetic Acid, Esters and Phenylacetonitriles
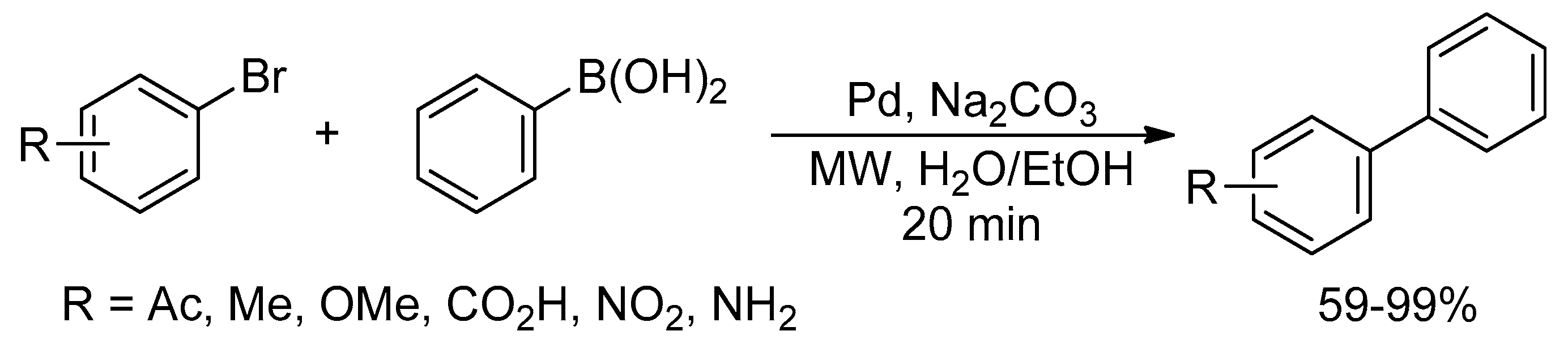
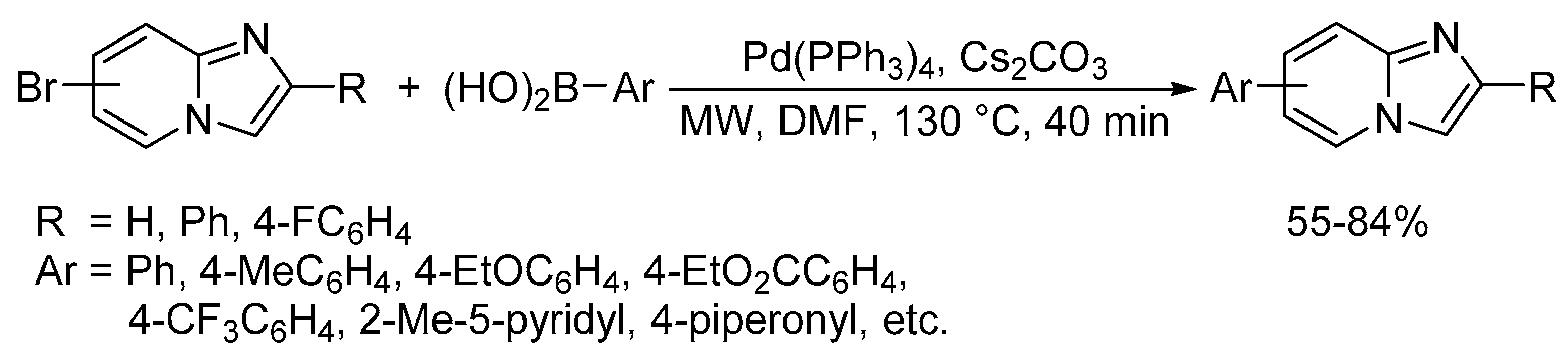
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
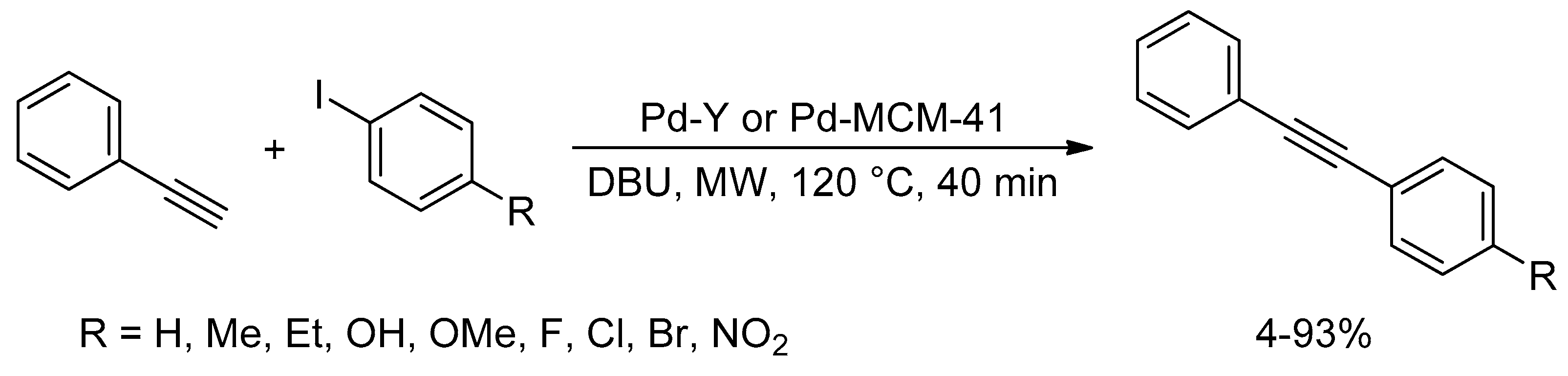
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
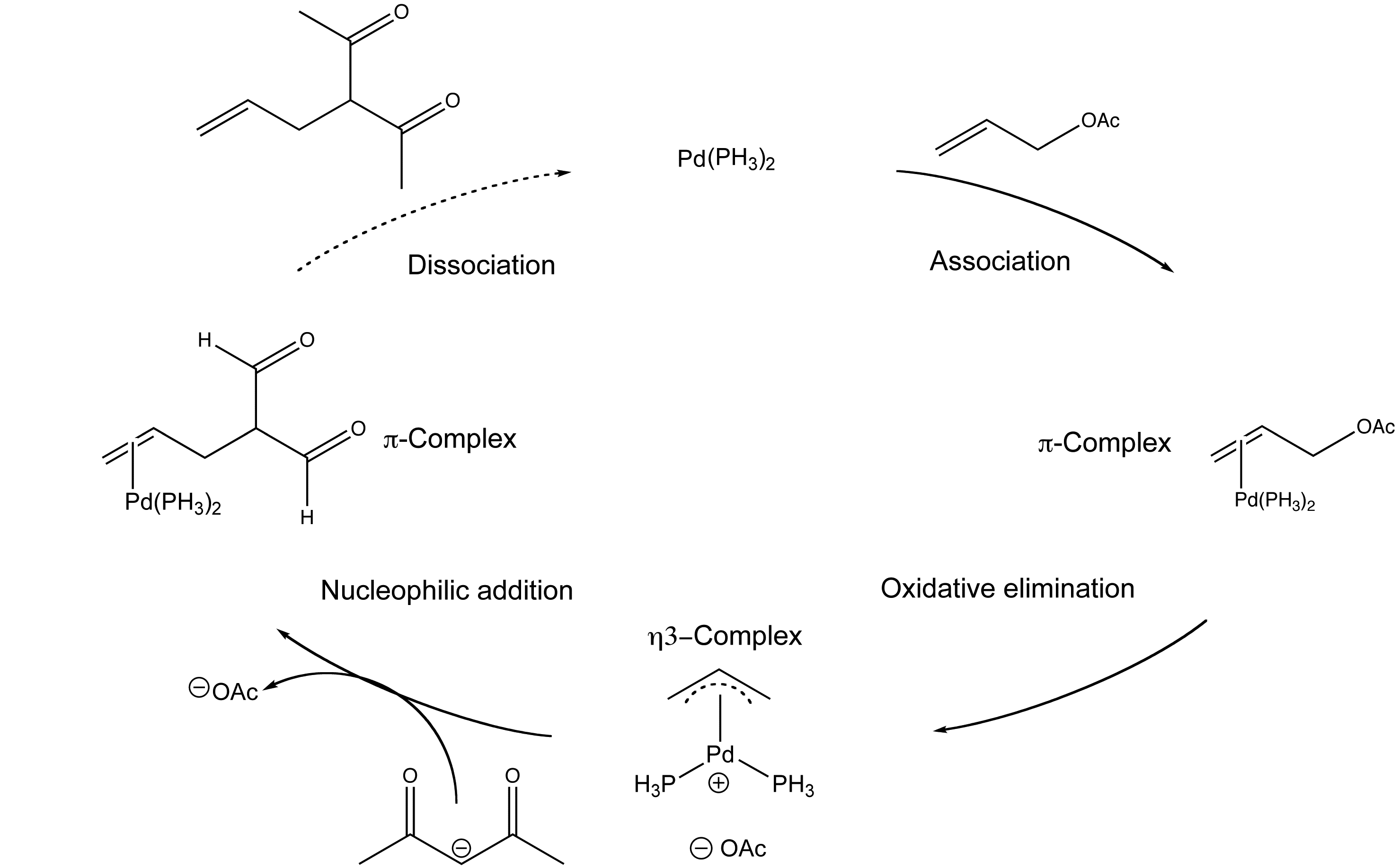
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

Direct synthesis of adipic acid esters via palladium-catalyzed carbonylation of 1,3-dienes | Science
![Palladium‐Catalyzed [4+2] and [5+2] Annulation for the Synthesis of Tetrahydroquinolines and 1,4‐Benzoxazepines - Eur. J. Org. Chem. - X-MOL Palladium‐Catalyzed [4+2] and [5+2] Annulation for the Synthesis of Tetrahydroquinolines and 1,4‐Benzoxazepines - Eur. J. Org. Chem. - X-MOL](https://xpic.x-mol.com/20200526%2F10.1002_ejoc.202000262.jpg)
Palladium‐Catalyzed [4+2] and [5+2] Annulation for the Synthesis of Tetrahydroquinolines and 1,4‐Benzoxazepines - Eur. J. Org. Chem. - X-MOL

Palladium-catalysed arylation of acetoacetate esters to yield 2-arylacetic acid esters | Semantic Scholar

Expanding the limit of Pd-catalyzed decarboxylative benzylations - Chemical Communications (RSC Publishing)

Figure 1 from Hydrodecarboxylation of Carboxylic and Malonic Acid Derivatives via Organic Photoredox Catalysis: Substrate Scope and Mechanistic Insight. | Semantic Scholar