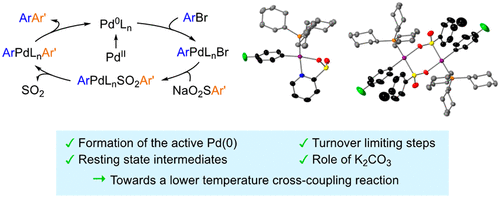
Mechanistic Studies of the Palladium-Catalyzed Desulfinative Cross-Coupling of Aryl Bromides and (Hetero)Aryl Sulfinate Salts. - J. Am. Chem. Soc. - X-MOL

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Palladium Nanoparticles Supported on Poly(N -vinylpyrrolidone)-Grafted Silica as an Efficient Catalyst for Copper-Free Sonogashira and Suzuki Cross-Coupling Reactions
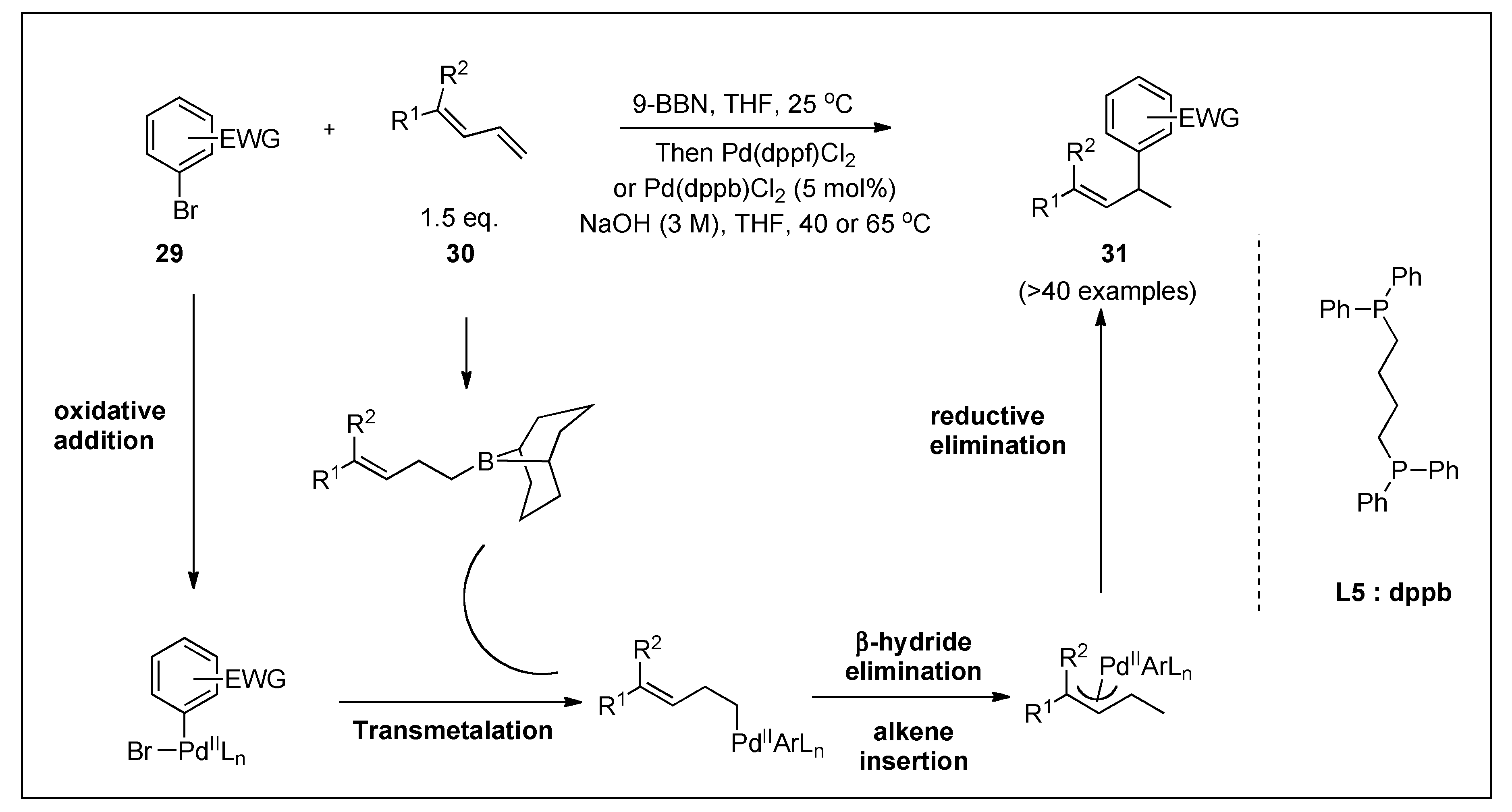
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML
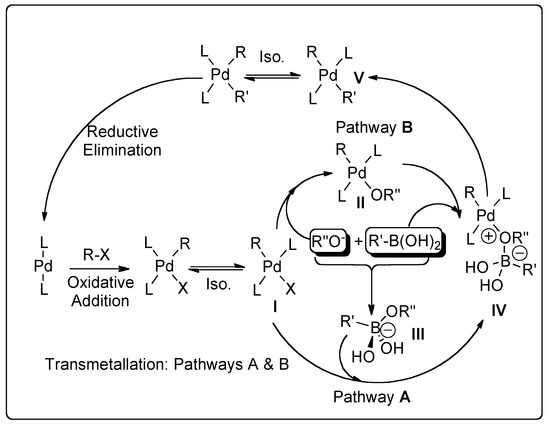
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML
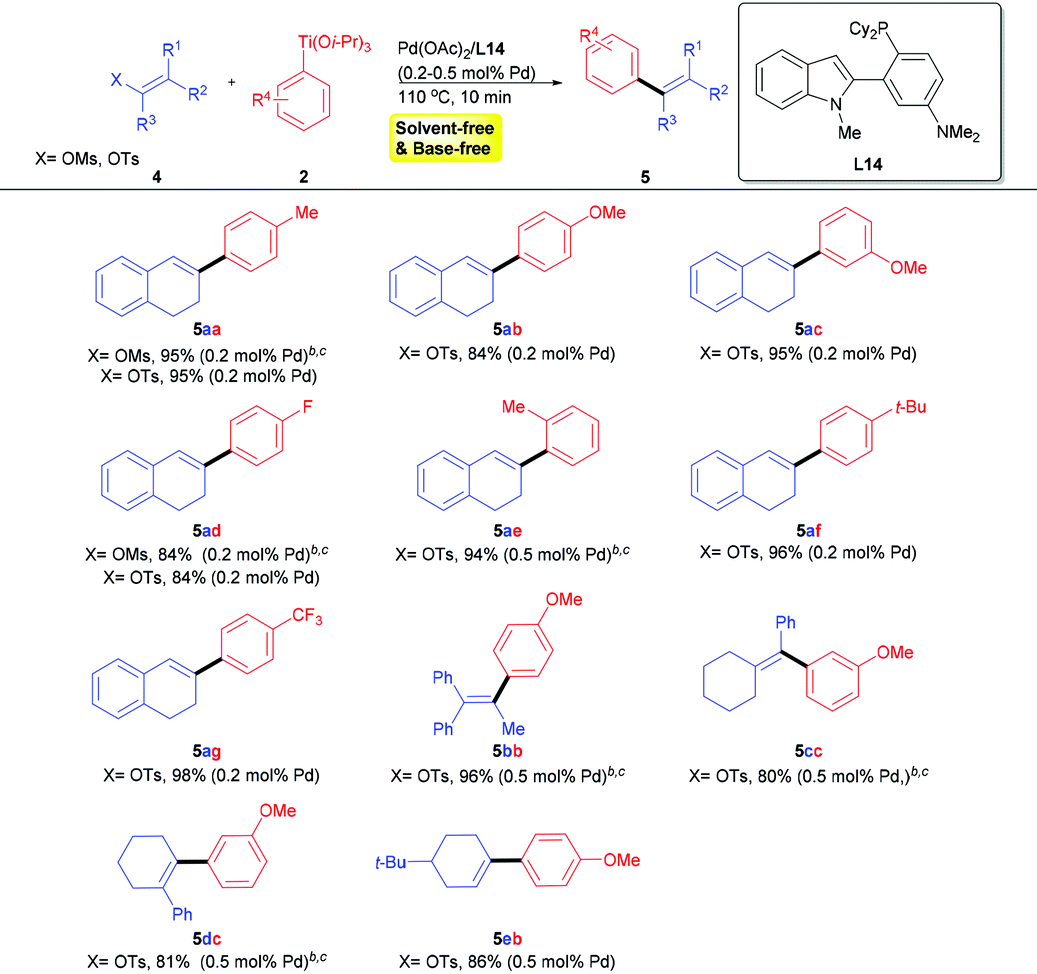
Palladium-catalyzed cross-coupling of (hetero)aryl or alkenyl sulfonates with aryl titanium as the multi-functional reagent - Organic Chemistry Frontiers (RSC Publishing)
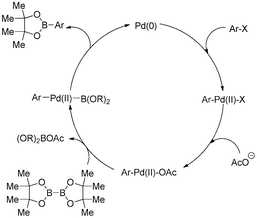
A decade advancement of transition metal-catalyzed borylation of aryl halides and sulfonates - RSC Advances (RSC Publishing)

A Pd-catalyzed, boron ester-mediated, reductive cross-coupling of two aryl halides to synthesize tricyclic biaryls - Organic & Biomolecular Chemistry (RSC Publishing)
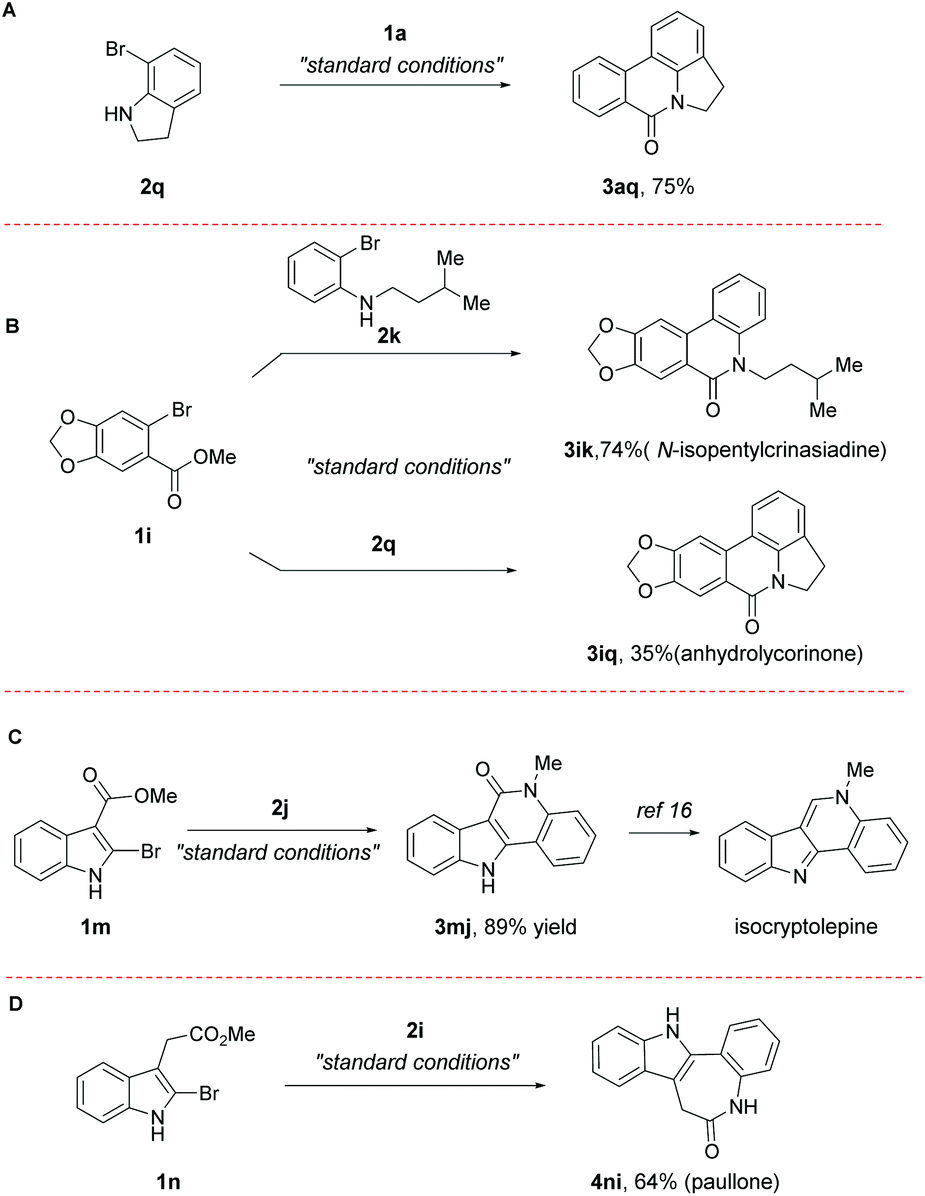
A Pd-catalyzed, boron ester-mediated, reductive cross-coupling of two aryl halides to synthesize tricyclic biaryls - Organic & Biomolecular Chemistry (RSC Publishing)

Regio- and stereoselective synthesis of tetra- and triarylethenes by N -methylimidodiacetyl boron-directed palladium-catalysed three-component coupling | Communications Chemistry

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
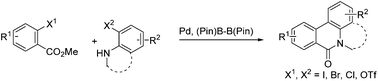
A Pd-catalyzed, boron ester-mediated, reductive cross-coupling of two aryl halides to synthesize tricyclic biaryls - Organic & Biomolecular Chemistry (RSC Publishing)

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
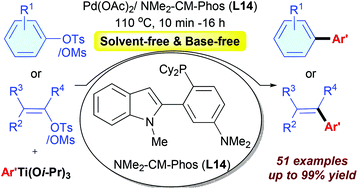
Palladium-catalyzed cross-coupling of (hetero)aryl or alkenyl sulfonates with aryl titanium as the multi-functional reagent - Organic Chemistry Frontiers (RSC Publishing)





